Standard-compliant software. Built simple.
Everything you need for ISO 13485 and 21 CFR Part 11. Without the corporate bloat.
Document Control
Automated workflows for creation, review, and e-signatures. Full audit trails included.
Risk Management
ISO 14971 compliant hazard analysis. Link risks directly to your design requirements.
Personnel Training
Automated training matrix. Ensure every team member is qualified for their tasks.
Change Management
Engineering changes and deviations managed in one place. No more lost spreadsheets.
Audit Ledger
A permanent record of every action. Get ready for your next audit in minutes, not weeks.
Quality Events
CAPA and non-conformance tracking. Root-cause analysis that actually leads to fixes.
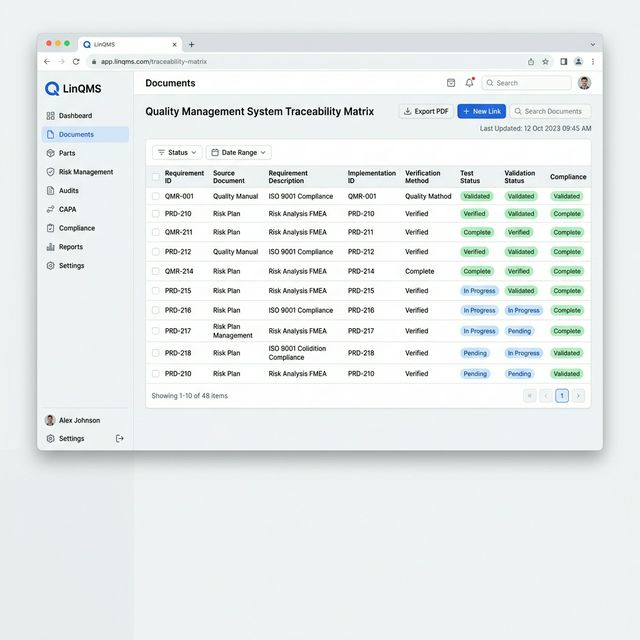
Automatic Traceability Matrix
LinQMS connects every record automatically. No manual mapping, no broken links.
Design Input
Hazard Analysis
Training Check
Release Order
Simple pricing
Early Access
LinQMS is currently in limited early access. Pricing will be finalized soon.
- Full Document Control
- Automated Traceability
- Personnel Training Matrix
- Audit-Ready Infrastructure
- Dedicated Onboarding Support
Regulatory Support
Our team of compliance experts is available 24/7 to help you configure your QMS for your specific regulatory needs.
Knowledge Base
Access templates, implementation guides, and best practices for ISO 13485 and FDA compliance.